Rapid Fire Session
Establishing T1 values for CMR at 0.55T

Christine Mancini, RT, FSCMR
CV MRI Technologist
National Heart, Lung, and Blood Institute, National Institutes of Health
Bethesda, Maryland, United States- SK
Sant Kumar
Fellow
National Heart, Lung, and Blood Institute, National Institutes of Health, United States - WB
W. Patricia Bandettini, MD
Senior Research Physician
National Heart, Lung, and Blood Institute, National Institutes of Health
Bethesda, Maryland, United States - JH
Jennifer Henry
The National Institutes of Health, United States
- ML
Margaret Lowery, RN
National Heart, Lung and Blood Institute (NHLBI), United States
- SB
Scott Baute
Physician Assistant
National Heart, Lung, and Blood Institute, National Institutes of Health, United States - AT
Anastasia Tsakirellis
Nurse Practitioner
National Institutes of Health, United States 
Adrienne E E. Campbell-Washburn, PhD
Principal Investigator
National Heart, Lung, and Blood Institute, National Institutes of Health
Bethesda, Maryland, United States
Presenting Author(s)
Primary Author(s)
Co-Author(s)
Methods: CMR was performed in a group of patients referred for clinical assessment of various cardiovascular conditions and in a group of healthy volunteers. All patients agreed to participate in an IRB-approved research protocol. Some patients underwent both a 0.55T scan and a 1.5T scan with a 60-day window. The 0.55T scans were performed on either a prototype MAGNETOM Aera (Siemens Healthineers, Forcheim, Germany) or a MAGNETOM Free.Max (Siemens) with investigational high-performance gradients. All 1.5T scans were performed on the same MAGNETOM Aera system that was later upgraded into a SolaFit (Siemens).
All patients underwent MOLLI native T1 mapping in short-axis and/or long-axis views. Patients who agreed to contrast received 0.15 mmol/kg gadobutrol for each scan and underwent post-contrast T1 mapping. Regions of interest (ROIs) were drawn on the T1 maps in the myocardium and left ventricular blood pool. In patients with myocardial infarction, ROIs in both normal (remote) myocardium and infarction were measured.
Subjects were categorized into one of the following groups: 1. Healthy volunteer, 2. myocardial infarction (MI), 3. cardiomyopathy, 4. acute inflammatory process, 5. amyloid, 6. hepatic iron overload, 7. acute COVID-19 infection, 8. post-COVID, 9. other cardiovascular condition.
Results: 435 patients underwent 0.55T scans (61 healthy volunteer, 16 acute COVID, 199 post-COVID, 159 of the other categorized diagnoses), 254/435 received contrast, and 110/435 patients underwent a comparison 1.5T scan. Table 1 provides native and post-contrast T1 values from the myocardium and blood, and ECV values, for 0.55T and 1.5T. At 0.55T, the average native myocardial T1 in healthy volunteers was 704ms, compared to 1028ms at 1.5T. T1 values were strongly correlated between field strengths (Figure 1A), and ECV measurements corresponded well with Bland-Altman bias = 0.84% (Figure 1B).
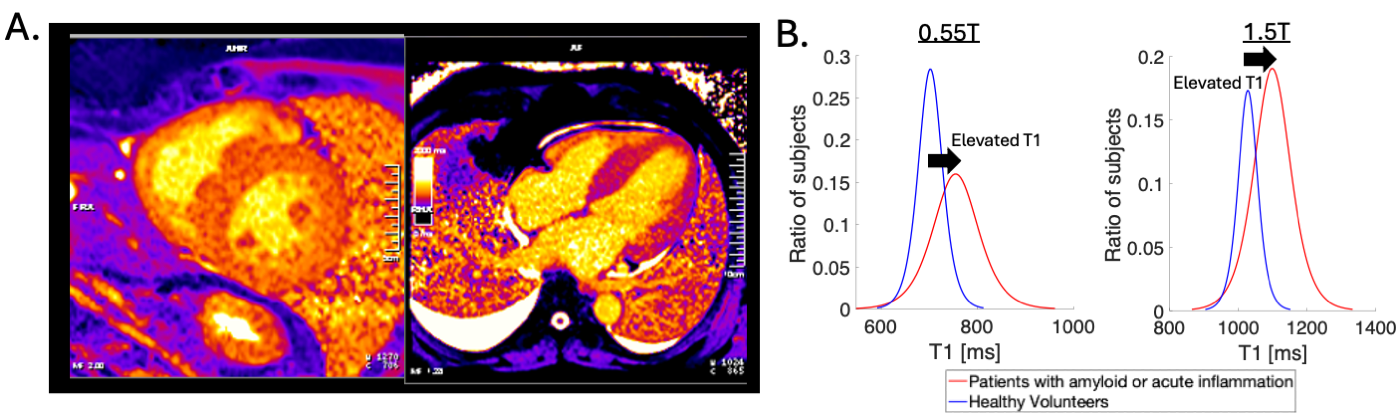
Importantly, elevated T1 indicative of clinically meaningful disease process was measurable at 0.55T (mean T1 >750ms, p< 0.05 by unpaired t-test versus healthy volunteers). Figure 2 provides example T1 maps in a patient with amyloidosis and histograms showing groups with higher T1 values at 0.55T and 1.5T.
Conclusion: Our study reports on normal T1 values at 0.55T and illustrates the application of 0.55T to detect elevated ECV and T1 in a clinically referred group of patients. This study adds to the growing literature supporting 0.55T CMR.
Table 1: T1 and ECV values measured at 0.55T and 1.5T.
| 0.55T T1 values (ms) | |||||
| Native | Post-contrast | ECV | |||
| Myocardium | Blood | Myocardium | Blood | ||
| Healthy volunteers | 704 ± 31 (61) | 1141 ± 79 (61) | 395 ± 82 (6) | 304 ± 101 (6) | 26 ± 4 (5) |
| MI (remote) | 705 ± 37 (34) | 1154 ± 63 (34) | 354 ± 38 (32) | 264 ± 30 (32) | 27 ± 5 (32) |
| MI (infarcted) | 559 ± 141 (25) | -- | 207 ± 69 (24) | -- | 64 ± 21 (24) |
| Cardiomyopathy | 717 ± 36 (19) | 1126 ± 102 (19) | 380 ± 71 (14) | 302 ± 154 (14) | 26 ± 5 (14) |
| Acute inflammatory process | 756 ± 69 (10) * | 1157 ± 113 (10) | 382 ± 47 (5) | 302 ± 82 (5) | 28 ± 6 (5) |
| Amyloid | 774 ± 26 (4) ** | 1147 ± 46 (4) | 253 ± 60 (3) | 265 ± 15 (3) | 54 ± 18 (3) |
| Hepatic iron overload | 731 ± 41 (43) | 1175 ± 83 (43) | -- | -- | -- |
| Acute COVID-19 | 719 ± 18 (16) | 1154 ± 82 (16) | 348 ± 16 (15) | 243 ± 25 (15) | 27 ± 3 (15) |
| Post-COVID-19 | 714 ± 22 (199) | 1123 ± 105 (197) | 349 ± 31 (185) | 240 ± 34 (184) | 26 ± 4 (184) |
| Other cardiovascular condition | 705 ± 28 (46) | 1145 ± 75 (46) | 362 ± 43 (30) | 256± 48 (30) | 24 ± 3 (30) |
| 1.5T T1 values (ms) | |||||
| Native | Post-contrast | ECV | |||
| Myocardium | Blood | Myocardium | Blood | ||
| Healthy volunteers | 1028 ± 34 (14) | 1592 ± 103 (14) | 544 ± 19 (3) | 413 ± 39 (3) | 27 ± 3 (3) |
| MI (remote) | 1027 ± 60 (33) | 1598 ± 31 (33) | 448 ± 68 (33) | 319 ± 30 (33) | 28 ± 6 (33) |
| MI (infarcted) | 948 ± 28 (24) | -- | 272 ± 78 (24) | -- | 61 ± 18 (24) |
| Cardiomyopathy | 1029 ± 51 (19) | 1520 ± 136 (19) | 439 ± 39 (15) | 306 ± 38 (14) | 26 ± 4 (14) |
| Acute inflammatory process | 1093 ± 74 (9) * | 1676 ± 132 (9) | 441 ± 56 (9) | 320 ± 80 (9) | 32 ± 7 (9) |
| Amyloid | 1139 ± 50 (3) * | 1607 ± 43 (3) | 296 ± 68 (3) | 303 ± 21 (3) | 54 ± 18 (3) |
| Hepatic iron overload | 1012 ± 57 (43) | 1572 ± 100 (43) | -- | -- | -- |
| Acute COVID-19 | -- | -- | -- | -- | -- |
| Post-COVID-19 | 998 ± 23 (5) | 1498 ± 79 (5) | 450 ± 18 (5) | 287 ± 17 (5) | 24 ± 2 (5) |
| Other cardiovascular conditions | 1027 ± 32 (40) | 1608 ± 87 (39) | 453 ± 34 (26) | 298 ± 39 (26) | 25 ± 2 (26) |
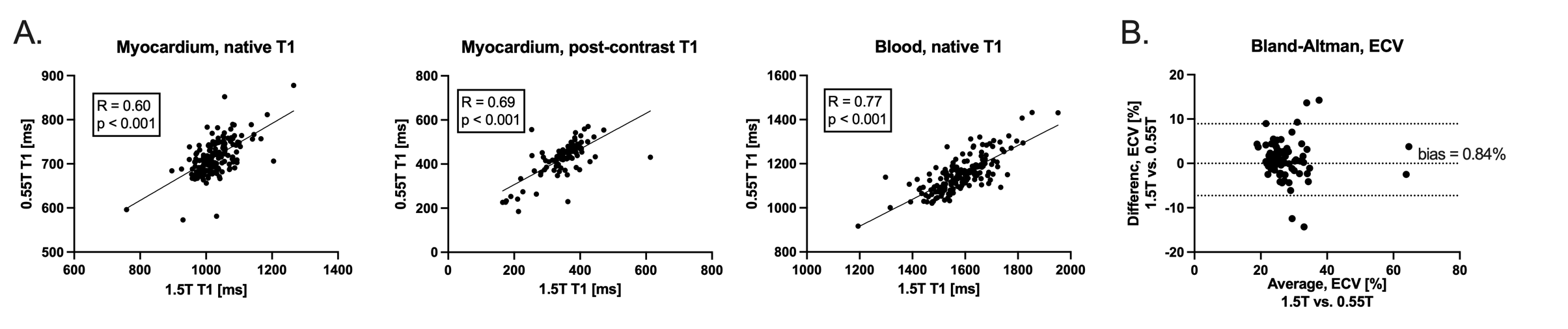 Figure 2: A) T1 maps from a patient presenting with severe left ventricular hypertrophy and exertional dyspnea. At 0.55T, myocardial native T1 values were increased at 760-790 ms (vs normal 704ms) and the extracellular volume fraction was 64%. The patient was confirmed to have cardiac amyloidosis. B) Histograms (with logistic fit) of T1 values to demonstrate elevated T1 in patients with amyloid or acute inflammation.
Figure 2: A) T1 maps from a patient presenting with severe left ventricular hypertrophy and exertional dyspnea. At 0.55T, myocardial native T1 values were increased at 760-790 ms (vs normal 704ms) and the extracellular volume fraction was 64%. The patient was confirmed to have cardiac amyloidosis. B) Histograms (with logistic fit) of T1 values to demonstrate elevated T1 in patients with amyloid or acute inflammation.