Rapid Fire Session
Adapting a CMR foundation model for A.I.-powered analysis of perfusion CMR: Enabling 12-fold reduction in manually labeled training dataset for automatic segmentation

M. Berk Sahin
PhD student
Purdue University
West Lafayette, Indiana, United States
M. Berk Sahin
PhD student
Purdue University
West Lafayette, Indiana, United States
Zhuoan Li, MSc
PhD Candidate
Laboratory for Translational Imaging for Microcirculation, Weldon School of Biomedical Engineering, Purdue University
Indianapolis, Indiana, United States
Khalid Youssef, PhD, MSc
Assistant Professor
Indiana University, Department of Radiology and Imaging Sciences
Indianapolis, Indiana, United States- AM
Arian M. Sohi, BSc
PhD Student
Laboratory for Translational Imaging for Microcirculation, Weldon School of Biomedical Engineering, Purdue University
Indianapolis, Indiana, United States - DY
Dilek M. Yalcinkaya, MSc
PhD Candidate
Purdue University
West Lafayette, Indiana, United States - LZ
Luis Zamudio, BSc
Research Engineer
Purdue University
Indianapolis, Indiana, United States 
Michael D. Elliott, MD
Director of Cardiac MRI
Atrium Health
Charlotte, North Carolina, United States
Venkateshwar Polsani, MD
Director of Cardiovascular Imaging
Piedmont Heart Institute
Atlanta, Georgia, United States- MT
Matthew S. Tong, DO
Associate Professor - Clinical
The Ohio State University
Columbus, Ohio, United States 
Dipan J. Shah, MD
Chief, Division of Cardiovascular Imaging Director, Cardiovascular MRI Laboratory
Weill Cornell Medical College, Houston Methodist DeBakey Heart and Vascular Center, Houston, Texas, USA
Houston, Texas, United States
Orlando P. Simonetti, PhD, FSCMR
Professor
The Ohio State University
Columbus, Ohio, United States- AH
Abolfazl Hashemi, PhD, MSc, BSc
Assistant Professor
Purdue University, Indiana, United States - BS
Behzad Sharif, PhD
Associate Professor
Purdue University
Indianapolis, Indiana, United States
Presenting Author(s)
Primary Author(s)
Co-Author(s)
Methods: We fine-tuned the cine FM using parameters learned from cine-based training. Data were obtained from the SCMR Registry [4] across four different centers. Two patients from each center were used for fine-tuning, and one from each center for validation. We then evaluated the FM on the same four test sets with a total of n=101 patients. An overview of this training and evaluation pipeline is described in Fig. 1. We compared our method with state-of-the-art 3D U-Net segmentation model [4], trained on a separate manually contoured FPP dataset of n=120 patients, which includes 12-fold more manual contours than our approach. Segmentation accuracy was assessed using the Dice score [6] and Haussdorf distance [7], two widely used metrics for overlap and boundary agreement between manual contours and segmentations.
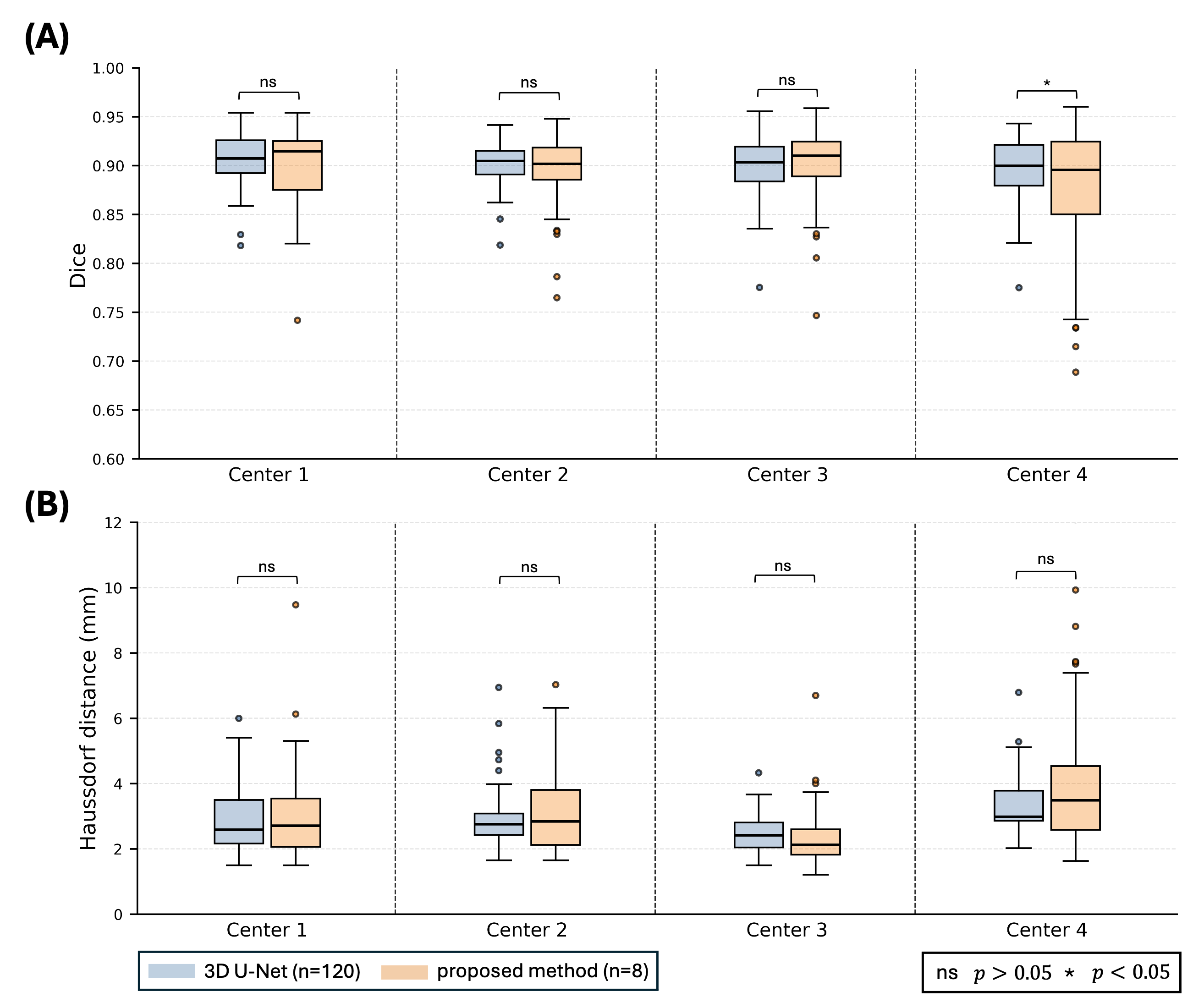
Results: With a 12-fold reduction in labeled training cases, the proposed method achieved performance comparable to the 3D U-Net trained on a much larger labeled dataset. Representative cases in Fig. 2 showed superior performance in RV and myocardial enhancement phases at center 2, with similar segmentations and Dice scores in other cases. Quantitative evaluation in Fig. 3 supported these findings. Differences in segmentation results between the proposed method and the 3D U-Net were not statistically significant (p > 0.05) across all centers for both metrics, except at center 4, where the Dice score showed a significant difference (p < 0.05).
Conclusion: Our results demonstrate that cine-based training on large unlabeled cine CMR data enables a FM to achieve state-of-the-art segmentation performance after fine-tuning with a 12-fold reduction in labeled training data. The method generalized robustly across centers, indicating that prior knowledge learned from cine CMR can be effectively transferred to FPP segmentation. This approach reduces reliance on extensive manual contouring, lessens the burden on clinicians, and supports scalable automated analysis of stress perfusion CMR.
Figure 1. Overview of the cine CMR foundation model training pipeline (A) and the proposed approach to adopt this model for myocardial segmentation of perfusion CMR data from the SCMR registry (B). The cine foundation model (FM) consists of four modules: a convolutional neural network (CNN) encoder extracts local features, a transformer encoder captures long-range dependencies between these features, a transformer decoder generates missing features, and a CNN decoder projects them back to the image space to construct the recovered image. We used cine FM trained on a large short-axis cine CMR dataset without manual contours using a masked image reconstruction task by minimizing the mean-squared error (MSE) loss, as described in panel (A) [3]. For FPP segmentation training, the CNN and transformer encoders were initialized with parameters learned from cine FM training and then fine-tuned, enabling transfer of the learned representations. A separate CNN decoder was used for the segmentation task. We trained cine FM on four SCMR multicenter datasets with limited manual contours to test whether prior cardiac structural knowledge from unlabeled cine FM traininng enables accurate FPP myocardial segmentation with only limited expert contours. From each center, 2 patients were used for training and 1 for validation, yielding a total of n=8 and n=4 patients, respectively. Fine-tuning was done by minimizing the sum of the cross-entropy (CE) loss and Dice loss [6] between the generated segmentations and manual contours as shown in panel (B). We evaluated the model on separate test sets from the same center 1-4 (Atrium Health, Piedmont, Houston Methodist, and Ohio State University), consisting of n=101 patients in total. For comparison, we evaluated a state-of-the-art 3D U-Net, which was trained on a separate substantially larger FPP dataset with manual contours (n=120 patients), on the same test sets [5].
.png) Figure 2: Representative cases from different centers showing the segmentations obtained with the 3D U-Net, the proposed method, and the corresponding manually drawn reference contours. For each center, the columns show (from left to right) the manually drawn contours followed by the automatic segmentations generated with the 3D U-Net and the proposed method, respectively. The number of patients with manual contours each method used for training or fine-tuning is indicated by “n”. Each row shows a different cardiac phase in FPP CMR: left ventricular (LV) enhancement, right ventricular (RV) enhancement, and myocardial enhancement. Dice score between the manual contour and the segmentation generated by the corresponding method is shown in yellow at the bottom-right corner of each perfusion image. The proposed method achieved segmentation accuracy comparable to the 3D U-Net, as reflected by Dice scores, despite being trained with substantially fewer manual contours. Importantly, in specific scenarios, such as the RV and myo. enhancement phase in center 2, it outperformed the 3D U-Net by a considerable margin.
Figure 2: Representative cases from different centers showing the segmentations obtained with the 3D U-Net, the proposed method, and the corresponding manually drawn reference contours. For each center, the columns show (from left to right) the manually drawn contours followed by the automatic segmentations generated with the 3D U-Net and the proposed method, respectively. The number of patients with manual contours each method used for training or fine-tuning is indicated by “n”. Each row shows a different cardiac phase in FPP CMR: left ventricular (LV) enhancement, right ventricular (RV) enhancement, and myocardial enhancement. Dice score between the manual contour and the segmentation generated by the corresponding method is shown in yellow at the bottom-right corner of each perfusion image. The proposed method achieved segmentation accuracy comparable to the 3D U-Net, as reflected by Dice scores, despite being trained with substantially fewer manual contours. Importantly, in specific scenarios, such as the RV and myo. enhancement phase in center 2, it outperformed the 3D U-Net by a considerable margin.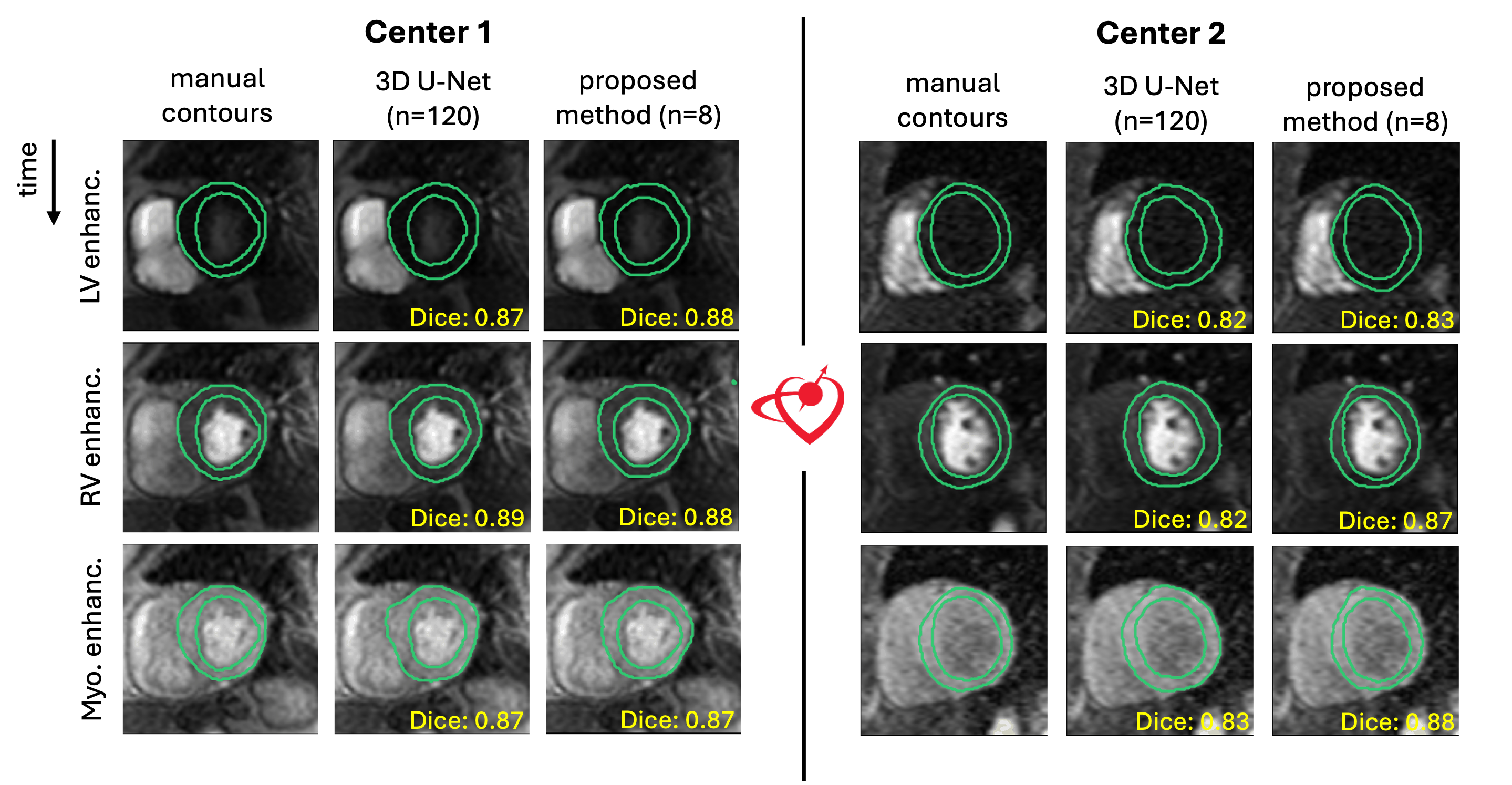 Figure 3: Cumulative first-pass perfusion CMR segmentation results across four centers, evaluated using different metrics. We quantitatively evaluated segmentation performance using Dice score [6], where higher values indicate better overlap, and Hausdorff distance [7], where lower values indicate better boundary agreement. Results for these metrics are shown in panels (A) and (B), respectively. Each panel is divided into four sections, corresponding to box-and-whisker plots for each center. Each box-and-whisker illustrates the segmentation performance of a method, with the method name specified in the legend at the bottom-left of the figure. The number of patients with manually drawn contours used for training each model is indicated by “n”. Statistical significance of the difference between 3D U-Net and the proposed method was calculated using the Student’s t-test. The statistical significance threshold for p value and the corresponding symbols are shown at the bottom-right of the figure. For both segmentation metrics, the difference between the 3D U-Net and the proposed method segmentations were not statistically significant (p > 0.05) at any center except center 4 with Dice score (p < 0.05).
Figure 3: Cumulative first-pass perfusion CMR segmentation results across four centers, evaluated using different metrics. We quantitatively evaluated segmentation performance using Dice score [6], where higher values indicate better overlap, and Hausdorff distance [7], where lower values indicate better boundary agreement. Results for these metrics are shown in panels (A) and (B), respectively. Each panel is divided into four sections, corresponding to box-and-whisker plots for each center. Each box-and-whisker illustrates the segmentation performance of a method, with the method name specified in the legend at the bottom-left of the figure. The number of patients with manually drawn contours used for training each model is indicated by “n”. Statistical significance of the difference between 3D U-Net and the proposed method was calculated using the Student’s t-test. The statistical significance threshold for p value and the corresponding symbols are shown at the bottom-right of the figure. For both segmentation metrics, the difference between the 3D U-Net and the proposed method segmentations were not statistically significant (p > 0.05) at any center except center 4 with Dice score (p < 0.05).